Describe How to Determine the Molar Mass of a Compound
Make use of the chemical formula to determine the number of atoms of each element in the compound. The final answer uses the correct number of significant figures.
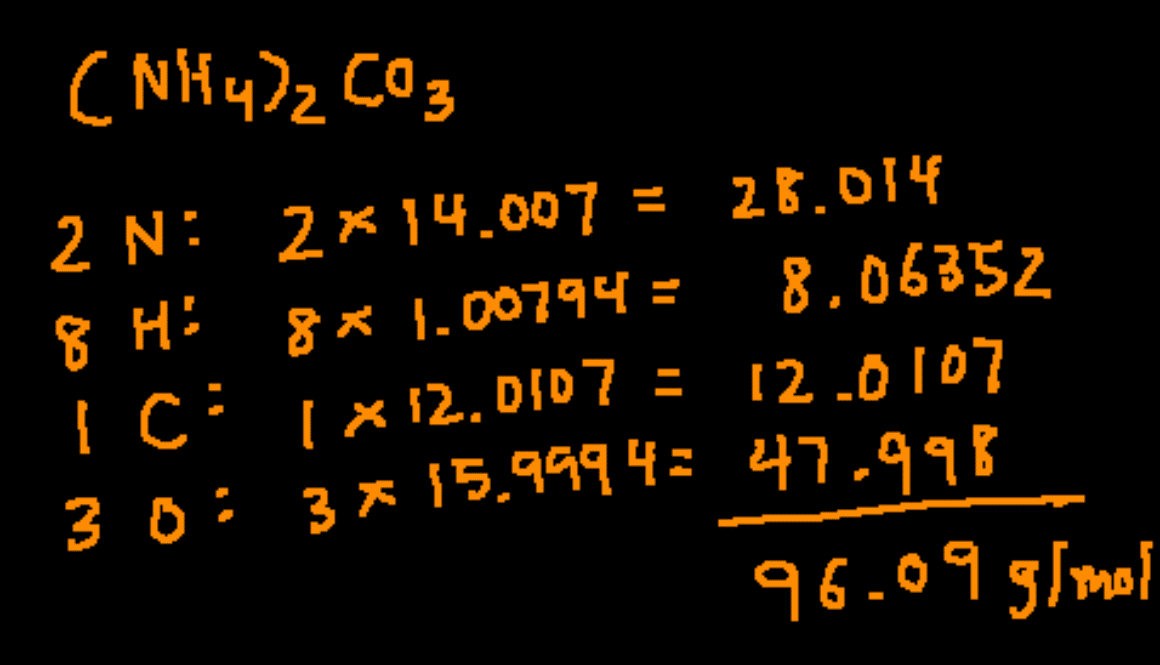
How To Calculate Molar Mass Step By Step With Examples
Use the freeing point depression to calculate the molality of the solution.
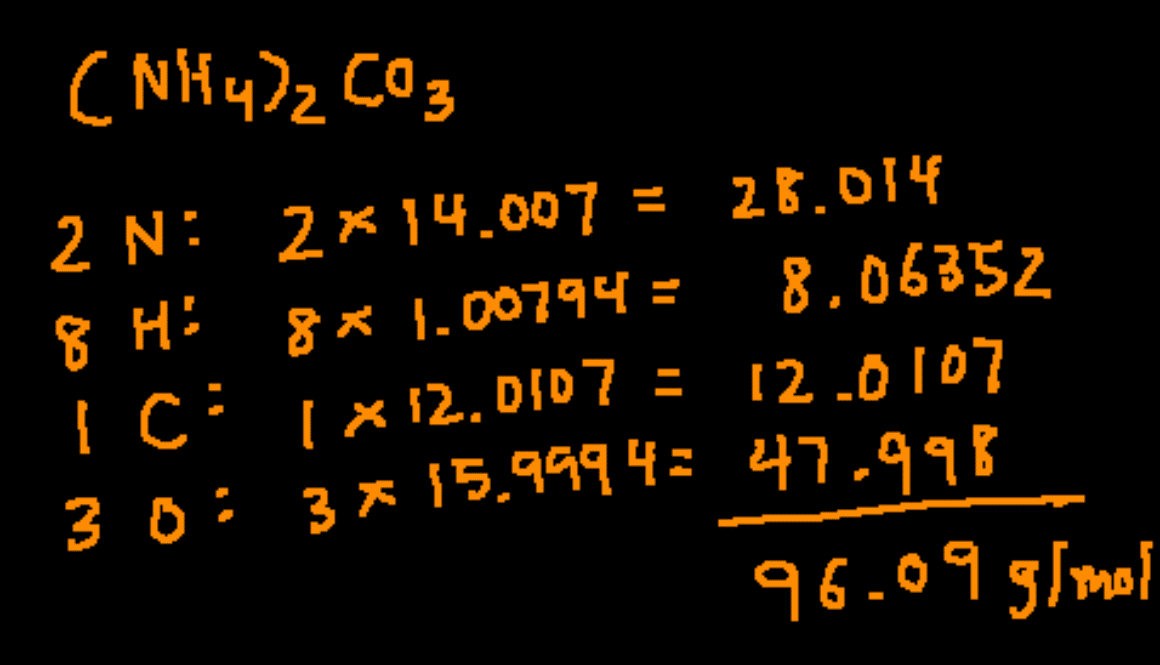
. Add the total and multiply it with. Molar mass solute. A brief lesson on finding the molar mass of a compound with three examples.
One mole of H 2 O is composed of 6023 x 10 23 H 2 O molecules. The molar mass is the combined atomic masses of the elements of the compound. If you read it correctly and rounded it to two decimal places you should get 2299.
In this case hydrogen has an atomic mass of 1 and oxygen has an atomic mass of 16. Molar mass of compound B is the sum of masses of 6023 x 10 23 B molecules. This chemistry video tutorial explains how to calculate the molar mass of a compound.
How do you calculate the molar mass. Multiply the mass of one mole of each element by the ratio of that element to one mole of the compound. Molar mass of compound A is the sum of masses of 6023 x 10 23 A molecules.
Finally add the products together and youll arrive at the answer. In this experiment you will determine the molar mass of an unknown solid by dissolving a pre-weighed. Multiply the atomic weight from the periodic table of each element by the number of atoms of that element present in the compound.
We then count the number of atoms and multiply it by the individual. Mass H 2 O 218 g 0218 kg. List the known quantities and plan the problem.
Thus molar mass of sodium Na is 2299 gmol. Calculate the molar mass of the solute. You can either memorize it or find all of the atomic masses located on the periodic table of elements.
The molar mass of a compound can be calculated by adding the standard atomic masses in gmol of the constituent atoms. In this compound there are 1 C 4 H 31 and 1 O. Therefore the units of molar mass are gramsmoleHow to find the molar mass of a compound.
K f is known as the freezing point depression constant and depends on the solvent used. Multiply the atomic weight of each element. Describe how to determine the molar mass of a compound.
Next multiply the number of a particular element by its molar mass. 101 Δ T f T f o T f K f m c. To calculate the Molar Mass of a compound first determine the atomic weight of each atom and then multiply the weight with the number of atoms in the compound.
The magnitude of the freezing depression produced by a solute is proportional to its colligative molality m c. The molar mass is the mass of a given chemical element or chemical compound g divided by the amount of substance mol. We can complete an example using the molecule Copper II Chloride.
Molar mass massmole gmol. This leads to two important facts. Determine the molal concentration m from the change in boiling point and the boiling point elevation constant.
Si 5 As 8. C 6 H 12 O 6. To find the molar mass find the atomic mass of all the components of a chemical.
Its molar mass is exactly 12 grams per mole. This is referred to as the gram formula mass or the molar mass. Determine the moles of unknown the solute from the molality of the solution and the mass of solvent in kilograms used to make the solution.
Therefore the molar mass of H. Then use the molality equation to calculate the moles of solute. After we have learned how to form these ionic compounds we can move on to calculating the mass of a compound.
To find the molar mass of a compound. Finding the molar mass of a compound is one of the most basic abilities a chemist. Grams per mol is another way of saying that for every 1 mole of Na there are 2299 g of sodium atoms in it.
Identify the conversion factors needed to convert between the number of moles and the mass of a compoun. The equation is therefore. Molecular mass 40078 x 3 3097361 x 2 159994 x 8 molecular mass 120234 6194722 1279952.
The total mass of 6023 x 10 23 H 2 O molecules is about 18 g. In other words the molar mass is the total mass of all the atoms in grams that make a mole of a particular molecule. It contains plenty of examples and practice problemsMy E-Book.
Copper II Chloride or CuCl2. In this case its five digits from the atomic mass for calcium. The definition of atomic mass the mole and molar mass are all directly or indirectly related to carbon-12.
Determine the molar mass from the mass of the unknown and the number of moles of unknown. Add the resulting masses. Now to get its molar mass we must attach the unit grams per mol to its atomic mass.
To determine the molar mass of a compound It is the addition of atomic masses of a compound. Use the periodic table. The first step in determining the molar mass is to determine the molecular mass of the compound which is weighted average mass of compounds naturally occurring molecules.
Mass solute 387 g. The mass of one atom of carbon-12 the atomic mass of carbon-12 is exactly 12 atomic mass units. To calculate the Molar Mass of a compound first determine the atomic weight of each atom and then multiply the weight with the number of atoms in the compound.
The mass of one mole of carbon-12 atoms is exactly 12 grams. Start by determining how many of each elements there are by looking the subscripts small number next to the element symbol. To calculate the molar mass of a molecule we first obtain the atomic weights from the individual elements in a periodic table.
Molecular mass 31017642 from the calculator molecular mass 31018. There is one atom of copper for every two atoms of chlorine in this molecule. Now we can apply this for real substances.
Give the molar mass of the compounds or molecules. Add it all together and put units of gramsmole after the number. 1 2 16 1 18.
Use the chemical formula to determine the number of each type of atom present in the compound.

How To Calculate The Molar Mass Of A Compound Quick Easy Youtube

How To Calculate Molar Mass Step By Step With Examples

How To Find Molar Mass Different Methods Of Calculation Explained With Examples
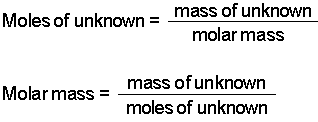
Comments
Post a Comment